COVID: Know Thine Enemy—-Molecular Considerations
My Childhood Obsessions:
I’ve always been interested in understanding things at a molecular level. I was a nerdy sort of a kid. Even at primary school I was interested in biology and chemistry. Fortunately, all my friends were pretty nerdy too, although I think I was the only one that had a hand-drawn “poster” of The Periodic Table of the Elements on his bedroom wall! At secondary school I decided that I wanted to do medicine, but when I applied to university my fallback position (should I not get into one of the Scottish medical schools) was to study biochemistry. As it turned out, two years into my medical course I got the opportunity to do an extra 2 years in the Science Faculty to get an intercalated honours degree in biochemistry. I then carried on to complete the last 3 years of my medical degree course.
The Renin-Angiotensin System:
I first came across the Renin-Angiotensin System (RAS) when I was studying physiology as a medical student, but I became more interested in the subject as a research registrar in the Medical Research Council Blood Pressure Unit in the 1980s. Renin, an enzyme produced in the kidneys, catalyses the conversion of the precursor peptide (small protein) angiotensinogen, which is produced in the liver, to the inactive Angiotensin I. Angiotensin I is then converted to Angiotensin II in a reaction catalysed by an enzyme called (unsurprisingly) Angiotensin Converting Enzyme (ACE). Angiotensin II binds to receptors in various tissues, and has a major role in blood pressure control by causing, among other effects, constriction of blood vessels and release from the adrenal glands of aldosterone (which in turn causes salt and water retention).
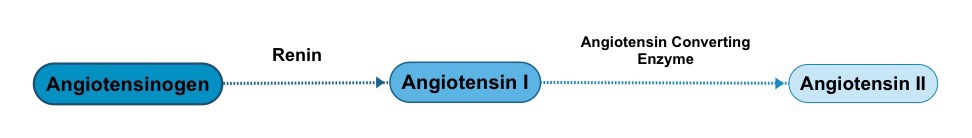
Drugs were developed to interfere with this system at 2 points: ACE inhibitors, which interfere with the production of Angiotensin II, and Angiotensin Receptor Blockers (ARBs), which interfere with the interaction between Angiotensin II and its receptors. These drugs are now major treatments for hypertension and heart failure.
It has been known for decades that locally expressed renin-angiotensin systems also exist in a number of tissues including the kidneys, the adrenal glands, the heart, blood vessels and the nervous system. These have a variety of functions, including cardiovascular regulation. They appear to work in association with, and independently of, the systemic RAS.
Angiotensin Converting Enzyme 2 (ACE2) and COVID:
What wasn’t known in the 1980s was that there exists another enzyme, related to ACE, called Angiotensin Converting Enzyme 2 (ACE2) which is attached to the cell membranes of cells in the lungs, arteries, heart, kidney and intestines. ACE2 catalyses the conversation of Angiotensin II to another peptide called Angiotensin (1-7) which, in contrast to Angiotensin II, causes dilatation of blood vessels. Hence, ACE2 counters the activity of the Renin-Angiotensin System.

When I started to read about COVID-19, one of the things that intrigued me about the responsible virus is that, like the previous Coronaviruses HCoV-NL63 and and SARS-CoV (the virus that causes SARS), SARS-CoV-2 uses ACE2 as its main entry point into cells. In order for the virus to enter a host cell, its spike (S) protein needs to be cleaved at 2 sites (S protein priming). This is catalysed by the host enzyme TMPRSS2, the inhibition of which might be a possible treatment for coronavirus infection.
Both ACE inhibitors and ARBs, because they interfere with the action of Angiotensin II, have been shown in animal models to increase production of ACE2. It was postulated that this might mean that patients treated with these drugs would be at higher risk from COVID-19. There is no conclusive evidence of this, however, so far and currently patients with COVID taking these drugs are advised not to change their treatment.
Apart from TMPRSS2 inhibition, another possible therapeutic avenue for COVID treatment is the use of anti-ACE2 antibodies to block the interaction between SARS-CoV-2 and ACE2.
A Final Thought:
The final thing that intrigues me about the story above is why these coronaviruses have evolved to attach to ACE2. It is reckoned that about a third of all human proteins are membrane proteins. In terms of evolution, what is the selective advantage that attaching to ACE2 gives to these viruses, as opposed to attaching to one of the many other membrane proteins?


This could have been a great power point for clinical forum Gordon! Really interesting read. In answer to your questions: I’m in the “dunno” camp.
LikeLike